Finding an average of 34.7 grams of salt per litre of seawater is no mystery: since the dawn of time, the sea has been a large body of salt water!
The oceans were formed about 4.2 billion years ago by rainwater running off the continent. As the water flowed across the continent, it carried with it salts of mineral origin, giving the sea its salty character.
The term salinity refers to the amount of salt in the water. In the ocean, the two main salts present are sodium (Na+) and chloride (Cl-), which combine to become sodium chloride (NaCl). This is the same salt that we find in our table salt shaker!
Today, 96.5% of seawater is pure water and 3.5% is made up of other substances, including salts. The amount of salt in the ocean is based on a balance between the evaporation of fresh water (caused by the wind at the surface) and the input of fresh water (through rainfall or runoff). The salts and minerals, too heavy to evaporate, remain in the ocean.
The two seas that produce the saltiest water in the world are the Mediterranean and the Red Sea, partly because of their semi-enclosed nature.
In contrast, the Black Sea has low salt content because it experiences more rainfall and runoff than evaporation of water. The polar regions are also areas of low salinity, due to the seasonal melting of ice in the Arctic Ocean.
The average salinity of the ocean is 34.7 psu (practical salinity unit), which is close to the salt content generally found in deep waters. At the surface, salinity often varies between 30 and 38 psu, depending on freshwater flows.
The study of salinity is important in oceanography, as is the study of temperature, to enable scientists to understand the movements of different water masses and thus understand the evolution of the ocean.
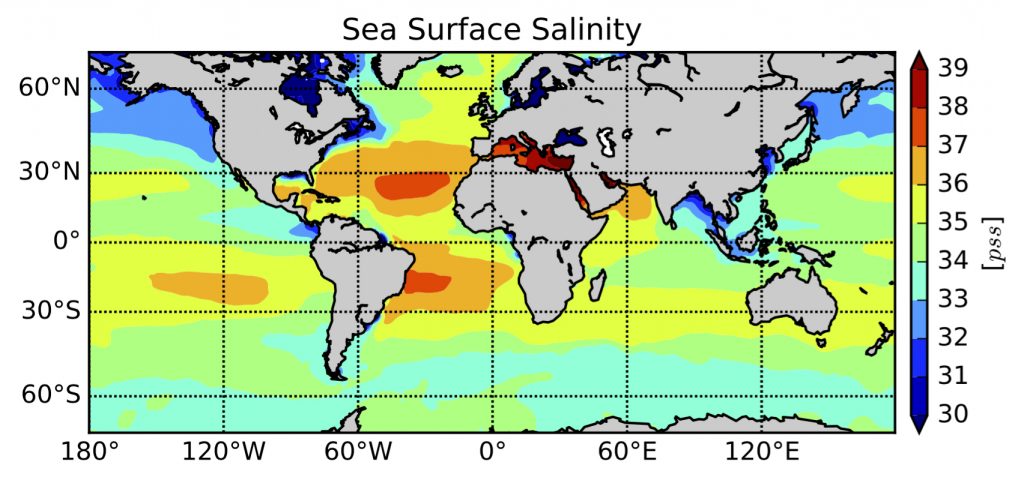
Global map of ocean salinity based on data collected at sea. Source : ISAS-13


